
Water is essential in cell biology – it possesses unique properties that make life possible. Three key properties of water are particularly relevant and are described in the scientific literature:
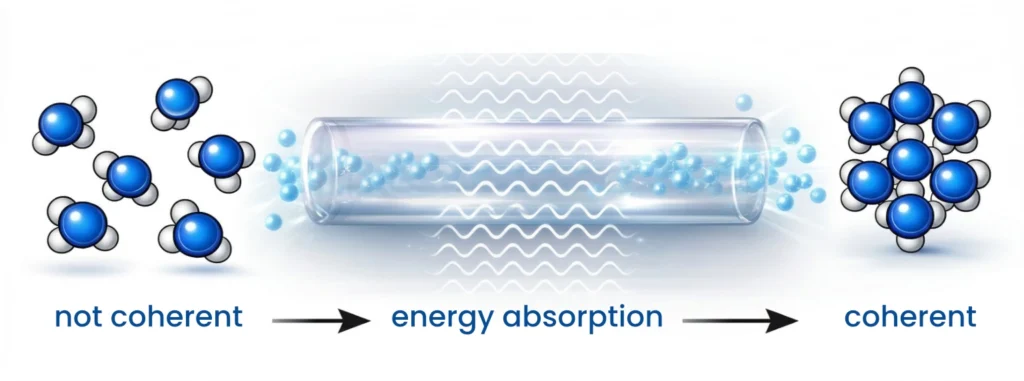
When water molecules absorb certain electromagnetic energies, coherent domains form; these are tiny regions of
increased order (low entropy). These are rapidly transmitted to the surrounding
water molecules, similar to a wave in a stadium. When these domains encounter a surface, they form a thin layer of ordered water molecules, the
so-called exclusion zone (EZ). Without a constant supply of coherent domains, these EZs are short-lived and cannot be stored. However, they play a crucial role in biological processes.
According to the quantum electrodynamics (QED) theory of water (developed by Del Giudice, Preparata, and Vitiello), coherent domains (CDs) form in liquid water under the influence of electromagnetic fields (EMF).
The generation of these domains does not occur through a simple chemical reaction, but rather through physical processes that influence molecular arrangement and vibration. Coherent domains represent a state of increased local order in water, induced by interaction with interfaces and electromagnetic fields. This phenomenon is central to explaining the unique physical properties of water in biological systems.
In cell biology, the properties of water—energy absorption, the formation of coherent domains, and surface order—are fundamental to protein folding. The process begins with the release of specific, absorbable electromagnetic energy by excited free radicals. This energy is absorbed by the surrounding water molecules and forms coherent domains, which create a thin layer of more highly ordered water molecules on the surface of unfolded proteins. Subsequently, the entropy exchange crucial for protein folding takes place. Unfolded proteins exist in a disordered state (high entropy). To fold into a more ordered protein, the unfolded protein must transition to an ordered state (low entropy). The water relinquishes its order, the proteins gain order, fold, and become structured and functional.
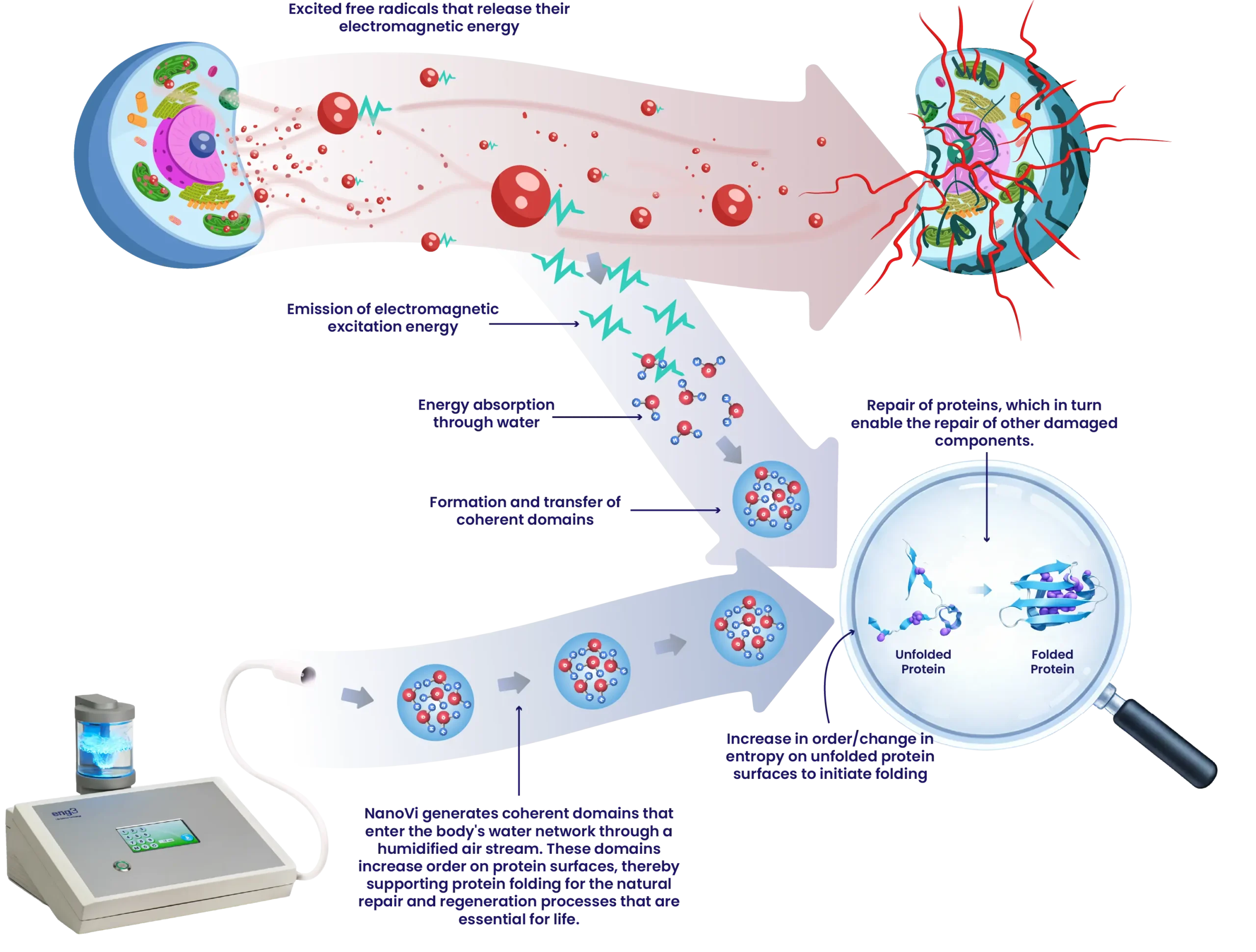
Protein folding is a thermodynamic process controlled by a change in entropy between the protein and the thin layer of ordered water surrounding it. Functional proteins are formed when an unfolded amino acid chain transitions from a state of low order (high entropy) to a state of high order (low entropy).
Ordered water, also called exclusion zone water (EZ water), has low entropy because its molecules are highly structured. In contrast, an unfolded amino acid chain has high entropy because it is disordered.
When an unfolded amino acid chain is surrounded by ordered water with low entropy, a change in entropy can occur. This transition causes the order in the water layers around the protein to decrease, allowing the protein to fold into its highly ordered, functional 3D structure. This mechanism demonstrates how crucial the correct water structure is for efficient protein folding and, consequently, for cell function.
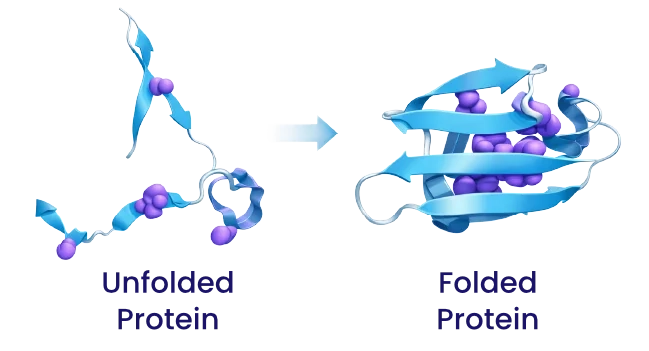
Protein folding is a complex thermodynamic process that requires and involves an entropy change (a change in entropy). The process is a fascinating example of the interplay between entropy and enthalpy in a biological system.
The net entropy change during protein folding is negative, meaning the system becomes more ordered. This seems to contradict the Second Law of Thermodynamics, which states that entropy must increase in an isolated system. However, protein folding is not an isolated system; it takes place in aqueous solution.
The overall process is described by the Gibbs-Helmholtz criterion:
ΔG = ΔH – TΔS
For folding to occur spontaneously, the Gibbs free energy (ΔG) must be negative.
The entropy contribution (ΔS) can be divided into two main components:
The total entropy change (ΔStotal) for the system (protein + water) is composed of the negative contribution of the protein and the positive contribution of the water.
ΔStotal = ΔSprotein + ΔSwater
The positive entropy gain from the solution water is usually large enough to offset or even exceed the negative entropy loss of the protein.
The hydrophobic effect is therefore the main driving force for protein folding, driven by the entropy gain of the water.